Fish reproductive biology and biotechnology
Fish Physiology and Genomics laboratory
Institut National de la Recherche Agronomique (INRA)
Rennes, France
Contact
Dr. Julien Bobe
Julien.bobe@rennes.inra.fr
Phone: +33 2 23 48 57 24
Dr. Catherine Labbé
Catherine.labbé@rennes.inra.fr
Phone: +33 2 23 48 50 04
Address
INRA LPGP
Campus de Beaulieu
35042 Rennes Cedex
FRANCE
Group description
The INRA Fish Physiology and Genomics laboratory harbors different research groups including a Sex Differentiation and Oogenesis group led by J Bobe and a Cell Cryoconservation and Fish Regeneration group led by C Labbé.
One of the applied objectives of the research developed within the Sex Differentiation and Oogenesis is to better control of egg quality in the context of new aquaculture systems and diversification of aquaculture species. Our group develops a functional genomic approach with a special interest for gene evolution in teleost fish and with the objective of better understanding the molecular mechanisms controlling egg quality. Our research is mainly focused on the contribution of maternally-inherited factors (e.g. maternal mRNAs) to the early developmental success.
The objective of the Cell Cryoconservation and Fish Regeneration group is to develop biotechnologies for preservation of aquatic species, in the perspective of genetic resources cryobanking and strain restoration. This includes the cryopreservation of sperm, embryonic cells, somatic cells and larvae in fish and mollusks, and fish regeneration from the cryopreserved cells, with a special interest for somatic cell nuclear transfer. Our research is mainly focused on epigenetic alterations after cryopreservation and nuclear transfer, and on cellular reprogramming during embryo development.
Main lines of research
– Oogenesis and egg quality in fish: Oocyte development. Oocyte Maturation. Ovulation. Artificial fertilization. Quality of eggs and spawnings. Maternal mRNAs. Functional genomics. Microarrays and NGS. Knock-down analysis. Zebrafish. Rainbow trout.
– Cell Cryoconservation and Fish Regeneration: Cryobiology. Freeze-thawing-induced damages. DNA methylation in gametes and embryos. Somatic cell reprogramming. Nuclear transfer. Cryobanking of aquatic genetic resources. Fish. Mollusk.
Links:
INRA Fish Physiology and Genomics: informal description of group activities, projects, publications, etc. www.lpgp.inra.fr
Institut National de la Recherche Agronomique (INRA): www.inra.fr
Relevant publications
- Migaud, H, Bell G, Cabrita E, McAndrew B, Davie A, Bobe J, Herráez MP, Carrillo M. 2013. Broodstock management and gamete quality in temperate fish. Reviews in Aquaculture. Reviews in Aquaculture. 5(Suppl 1): 194-223.
- Chenais, N., Depince, A., Le Bail, P-Y., Labbé, C. (2013) Fin cell cryopreservation and fish reconstruction by nuclear transfer stand as promising technologies for preservation of finfish genetic resources. Aquaculture International. 22, 63-76.
- Marandel L, Labbé C, Bobe J, Le Bail PY. 2012. nanog 5′-upstream sequence, DNA methylation, and expression in gametes and early embryo reveal striking differences between teleosts and mammals. Gene. 492(1):130-7.
- Marandel L, Labbé C, Bobe J, Le Bail PY. 2012. c-myc evolutionary history in teleosts and duplicated c-myca genes characterization in goldfish embryos. Molecular Reproduction and Development. 79(2):85-96.
- Fauvel, C., Boryshpolets, S., Cosson, J., Wilson Leedy, J., Labbé, C., Haffray, P., Suquet, M. (2012). Improvement of chilled seabass sperm conservation using a cell culture medium. Journal of Applied Ichthyology, 28, 961-966.
- Suquet, M., Cosson, J., Donval, A., Labbé, C., Boulais, M., Haffray, P., Bernard, I., Fauvel, C. (2012). Marathon vs sprint racers: an adaptation of sperm characteristics to the reproductive strategy of pacific oyster, turbot and seabass. Journal of Applied Ichthyology, 28, 956-960.
- Marandel L, Labbe C, Bobe J, Jammes H, Lareyre JJ, Le Bail PY. 2013.Do not put all teleosts in one net: Focus on the sox2 and pou2 genes. Comp Biochem Physiol B Biochem Mol Biol. (164(2):69-79.
- Lubzens E, Young G, Bobe J, Cerdà J. 2010. Oogenesis in teleosts: how fish eggs are formed? General and Comparative Endocrinology. 165: 367-389.
- Desvignes T, Fauvel C, Bobe J 2011. The Nme gene family in zebrafish oogenesis and early development. NS Archives Pharmacology. 384(4-5):439-49.
- Crespo D, Planas J, Bobe J. 2010. Lipopolysaccharide administration induces premature ovulation and reduced egg quality in rainbow trout (Oncorhynchus mykiss). Aquaculture. 300: 240-242.
- Bobe, J., Labbé, C., 2009. Chilled storage of sperm and eggs. In “Methods in reproductive aquaculture: Marine and freshwater species”. CRC Press, Boca Raton (US) Collection Marine biology series, (6) 219-235
- Bonnet E, Fostier A, Bobe J 2007 Microarray-based analysis of fish egg quality after natural or controlled ovulation. BMC Genomics 8:55
- Bonnet E, Fostier A, Bobe J 2007 Characterization of rainbow trout egg quality: A case study using four different breeding protocols, with emphasis on the incidence of embryonic malformations. Theriogenology 67:786
- Bobe J, Labbé C. Egg and sperm quality in fish. 2010. General and Comparative Endocrinology. 165: 535-548.
- Aegerter S, Jalabert B, Bobe J 2005 Large scale real-time PCR analysis of mRNA abundance in rainbow trout eggs in relationship with egg quality and post-ovulatory ageing. Molecular Reproduction and Development 72:377-385
IFREMER
Contact
Dr. Christian Fauvel
cfauvel@ifremer.fr
Phone: +33 4 67 13 04 14
BOME : Chemin de Maguelone
34 250 Palavas-les-Flots, France
M. Suquet
msuquet@ifremer.fr
Phone: +33 2 98 89 29 49
LPI : Ifremer, Pointe du Diable
29280 Plouzané, France
Group description
IFREMER, the French Research Institute for the Exploitation of the Sea, is an industrial and commercial public company exclusively devoted to maritime interests. It operates under the joint auspices of three ministries (research and education, agriculture and fisheries and equipment).
IFREMER research on living marine resources are undertaken in different research units and facilities situated along French coast and overseas. Two main IFREMER structures are involved in gamete biology:
LPI (Laboratory of Invertebrate Physiology) in Brest and its satellite on seashore (station of Argenton), including 10 researchers and 2 post docs, 9 technicians and 6 PhD fellows, are dedicated to oyster and scallop physiology in relation to their environment. In this unit, mollusc sperm and eggs which have not been thoroughly described so far are analysed following a multifactorial approach with special reference to metabolism. The acquired knowledge is implemented to develop gamete and embryo banking of target species as well as improving hatchery based mollusc production.
BOME (Biology of Exploited Marine Organisms) housing 9 researchers, 7 technicians and 3PhD fellows, in Palavas is mainly dedicated to fish aquaculture development through sustainability, genetics and ecophysiology. In this unit, the determinism of gamete quality is investigated through broodstock husbandry and gamete management after collection in both sexes. The applied results are implemented through large factorial crosses for genetic studies and through improvement of gamete banking.
Both units lean upon universities and other national research bodies through local UMRs (common research units ) which give access to a large variety of approaches from simple gamete handling to molecular biologytools and skills. Moreover, tight relations with INRA LGP are maintained through national and international collaborations.
Main lines of research
– Gamete biology and quality: Sperm and oocyte metabolism, sperm movement, fertilization, egg and sperm quality, seabass, turbot, tuna, oyster, scallop.
– Gamete cryopreservation: Sperm and egg quality, cryopreservation, diluents, fertilization, larval rearing. Gamete energetics
Links:
Ifremer: http://wwz.ifremer.fr/institut_eng/
Ifremer Bome: http://wwz.ifremer.fr/mediterranee_eng/implantations/Palavas
Ifremer LPI/Brest: http://wwz.ifremer.fr/pfompi
Ifremer LPI/Argenton : http://www.ifremer.fr/argenton
Relevant publications
1. Desvignes T., Fostier A., Fauvel Christian, Bobe J. (2013). The Nme gene family in fish. Fish Physiology And Biochemistry, 39(1), 53-58. Publisher’s official version: http://dx.doi.org/10.1007/s10695-012-9639-8, Open Access version: http://archimer.ifremer.fr/doc/00133/24466/
2. Suquet Marc, Quere Claudie, Mingant Christian, Lebrun Luc, Ratiskol Dominique, Miner Philippe, Cosson Jacky (2013). Effect of sampling location, release technique and time after activation on the movement characteristics of scallop (Pecten maximus) sperm. Aquatic Living Resources, 26(3), 215-220. Publisher’s official version: http://dx.doi.org/10.1051/alr/2013048, Open Access version: http://archimer.ifremer.fr/doc/00157/26852/
3. Zupa R., Fauvel Christian, Mylonas C. C., Santamaria N., Valentini L., Pousis C., Papadaki M., Suquet Marc, De La Gandara F., Bello G., Metrio G., Corriero A. (2013). Comparative analysis of male germ cell proliferation and apoptosis in wild and captive Atlantic bluefin tuna (Thunnus thynnus L.). Journal Of Applied Ichthyology, 29(1), 71-81. Publisher’s official version: http://dx.doi.org/10.1111/j.1439-0426.2012.02045.x, Open Access version: http://archimer.ifremer.fr/doc/00125/23579/
4. Suquet Marc, Cosson J., Donval Anne, Labbe C., Boulais Myrina, Haffray Pierrick, Bernard Ismael, Fauvel Christian (2012). Marathon vs sprint racers: an adaptation of sperm characteristics to the reproductive strategy of Pacific oyster, turbot and seabass. Journal Of Applied Ichthyology, 28(6), 956-960. Publisher’s official version: http://dx.doi.org/10.1111/jai.12061, Open Access version: http://archimer.ifremer.fr/doc/00111/22220/
5. Fauvel Christian, Boryshpolets Sergey, Cosson J., Leedy J. G. Wilson, Labbe C., Haffray Pierrick, Suquet Marc (2012). Improvement of chilled seabass sperm conservation using a cell culture medium. Journal Of Applied Ichthyology, 28(6), 961-966. Publisher’s official version: http://dx.doi.org/10.1111/jai.12071, Open Access version: http://archimer.ifremer.fr/doc/00111/22221/
6. Corporeau Charlotte, Vanderplancke Gwenaelle, Boulais Myrina, Suquet Marc, Quere Claudie, Boudry Pierre, Huvet Arnaud, Madec Stephanie (2012). Proteomic identification of quality factors for oocytes in the Pacific oyster Crassostrea gigas. Journal Of Proteomics, 75(18), 5554-5563. Publisher’s official version: http://dx.doi.org/10.1016/j.jprot.2012.07.040, Open Access version: http://archimer.ifremer.fr/doc/00098/20925/
7. Suquet Marc, Le Mercier Alain, Rimond Flore, Mingant Christian, Haffray Pierrick, Labbe C. (2012). Setting tools for the early assessment of the quality of thawed Pacific oyster (Crassostrea gigas) D-larvae. Theriogenology, 78(2), 462-467. Publisher’s official version: http://dx.doi.org/10.1016/j.theriogenology.2012.02.014, Open Access version: http://archimer.ifremer.fr/doc/00088/19934/
8. Desvignes T., Fauvel Christian, Bobe J. (2011). The nme gene family in zebrafish oogenesis and early development. Naunyn-Sschmiedebergs Archives Of Pharmacology, 384(4-5), 439-449. Publisher’s official version: http://dx.doi.org/10.1007/s00210-011-0619-9, Open Access version: http://archimer.ifremer.fr/doc/00051/16226/
9. Desvignes Thomas, Fauvel Christian, Fostier Alexis, Bobe Julien (2011). NME family and determination of egg quality, new insights from the zebrafish Danio rerio. Indian Journal of Science and Technology, 4(S8), 214-215. Open Access version: http://archimer.ifremer.fr/doc/00049/16035/
10. Groison A. -L., Fauvel Christian, Suquet Marc, Kjesbu O. S., Le Coz Jean-Rene, Mayer I., Cosson J. (2010). Some characteristics of sperm motility in European hake (Merluccius merluccius, L., 1758). Journal Of Applied Ichthyology, 26(5), 682-689. Publisher’s official version: http://dx.doi.org/10.1111/j.1439-0426.2010.01541.x, Open Access version: http://archimer.ifremer.fr/doc/00015/12644/
11. Suquet Marc, Cosson J., De La Gandara F., Mylonas C. C., Papadaki M., Lallemant S., Fauvel Christian (2010). Sperm features of captive Atlantic bluefin tuna (Thunnus thynnus). Journal Of Applied Ichthyology, 26(5), 775-778. Publisher’s official version: http://dx.doi.org/10.1111/j.1439-0426.2010.01533.x , Open Access version: http://archimer.ifremer.fr/doc/00015/12645/
12. Cosson J., Groison , Fauvel Christian, Suquet Marc (2010). Description of hake (Merlucius merlucius) spermatozoa: flagellar wave characteristics and motility parameters in various situations. Journal Of Applied Ichthyology, 26(5), 644-652. Publisher’s official version: http://dx.doi.org/10.1111/j.1439-0426.2010.01563.x, Open Access version: http://archimer.ifremer.fr/doc/00015/12643/
13. Fauvel Christian, Suquet Marc, Cosson J. (2010). Evaluation of fish sperm quality. Journal Of Applied Ichthyology, 26(5), 636-643. Publisher’s official version: http://dx.doi.org/10.1111/j.1439-0426.2010.01529.x , Open Access version: http://archimer.ifremer.fr/doc/00015/12642/
14. Suquet Marc, Labbe C., Brizard Raphael, Donval Anne, Le Coz Jean-Rene, Quere Claudie, Haffray P. (2010). Changes in motility, ATP content, morphology and fertilisation capacity during the movement phase of tetraploid Pacific oyster (Crassostrea gigas) sperm. Theriogenology, 74(1), 111-117. Publisher’s official version: http://dx.doi.org/10.1016/j.theriogenology.2010.01.021 , Open Access version: http://archimer.ifremer.fr/doc/00006/11756/
15. Crespel Amélie, Rime Helene, Fraboulet Erwann, Bobe Julien, Fauvel Christian (2008). Egg quality in domesticated and wild seabass (Dicentrarchus labrax): A proteomic analysis. Cybium, Revue Internationale d’Ichtyologie, 32(2), 205. Open Access version: http://archimer.ifremer.fr/doc/00000/6555/
Research Unit Animal and Functionality of Animal Products (URAFPA)
Team Domestication in Inland Aquaculture (DAC)
Université de Lorraine (UL) / Institut National de la Recherche Agronomique (INRA)
Nancy, France
Contact
Pr. Pascal Fontaine
p.fontaine@univ-lorraine.fr
Phone: +33 3 83 68 56 00
Dr. Berenice Schaerlinger
Berenice.Schaerlinger@univ-lorraine.fr
Phone: +33 3 83 68 55 96
Address
URAFPA Team DAC, Faculté des Sciences et Technologies
Boulevard des Aiguillettes, BP 236
F-54506 Vandoeuvre-les-Nancy cedex
Group description
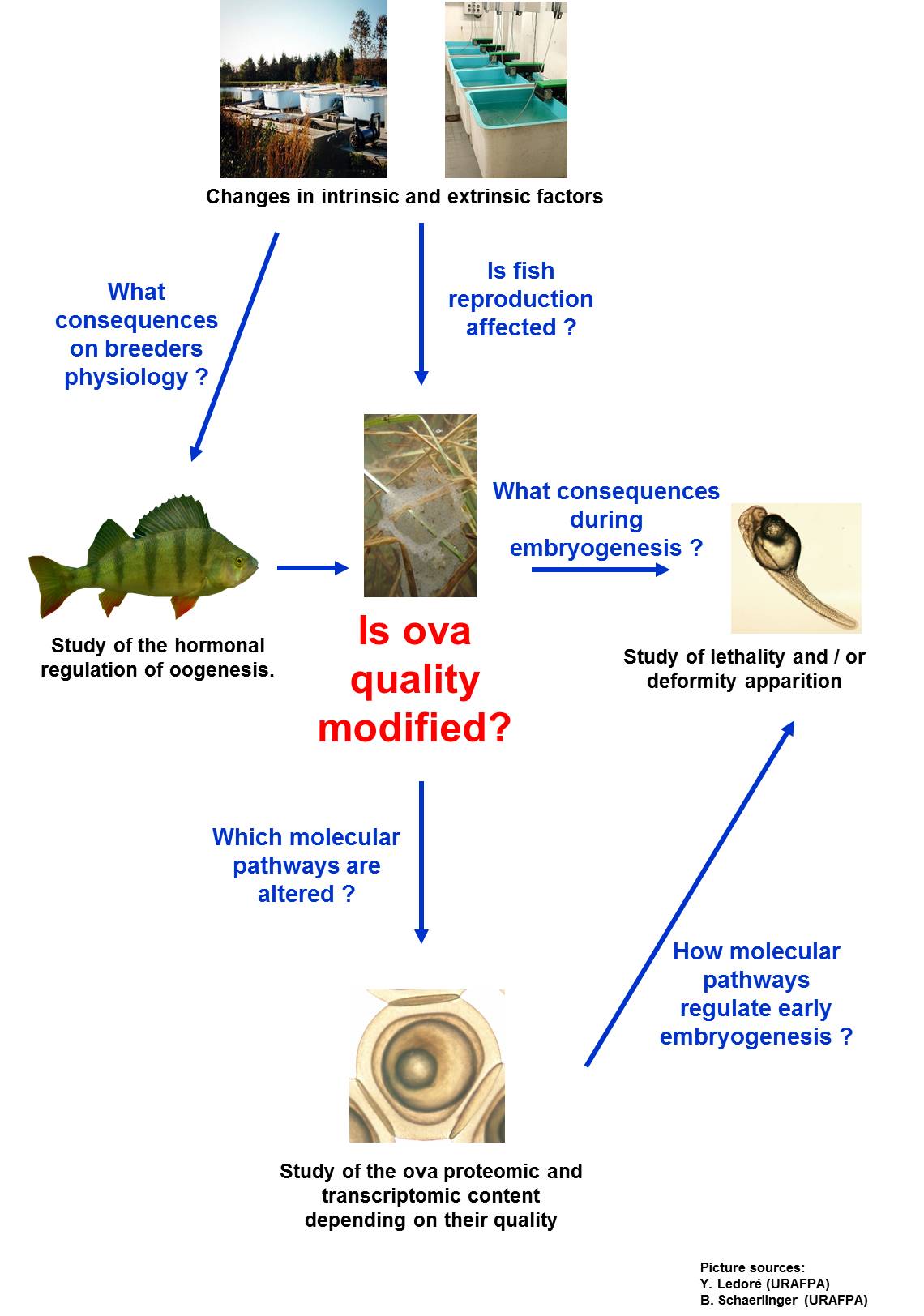
The Research Unit Animal and Functionality of Animal Products is composed of 4 teams among them the team Domestication in Inland Aquaculture (DAC) animated by P. Fontaine. This team develops a generic approach for the domestication of new species in fish farming and studies the effect of domestication process on freshwater fish reproductive biology and performance. The principal model organism is the Eurasian perch (Perca fluviatilis) for which the knowledge of the environmental control on the reproduction cycle is well known but the mechanisms underlying this control are still poorly understood. Moreover, experiments are also conducted on pike-perch (Sander lucioperca), Northern pike (Esox lucius), roach (Rutilus rutilus), rudd (Scardinius erythrophtalmus) ande burbot (Lota lota).
One research topic of DAC deals with the regulation of several physiological components of the brain-hypophysis-gonadic axis (neurotransmitter, hormones) in relation to the intrinsic and extrinsic factors controlling oogenesis (e.g. domestication, geographical origin, temperature, and photoperiod). Moreover, studies aiming to establish relationships between the changes of these physiological parameters and the oocyte development are performed to understand the control of maternally deposited/synthesized proteins and mRNA. The disruption of these processes may be linked to the ova quality and those molecules may constitute potential molecular indicators of gametes quality. (Collaboration with J. Bobe, INRA Rennes)
Another topic is to evaluate the ova quality through the research of new molecular indicators using proteomic and transcriptomic approaches. These techniques allow us to identify maternal pathways regulating key fonctions during fish early life stages. Indeed, the misregulation of these pathways leads to early lethality or deformities apparition during the embryogenesis. The links between these molecular pathways and the embryogenesis defects are investigated through microscopic and functional approaches to understand their importance for the proper development of embryos and larvae.
As a whole, those researches aim at understanding the links between the intrinsic factors or the environmental perception of breeders and the effect on the oogenesis, gametes quality and reproduction success to ensure the production of a large number of resistant progeny.
Main lines of research
– Eurasian perch, reproduction control, oogenesis, brain-hypophysis-gonadic axis, ova quality, proteomic, transcriptomic, molecular pathways, embryonic and larval development.
Links:
URAFPA: description of the team activities and publications. http://www.urafpa.fr
Relevant publications
1. Abdulfatah A., Fontaine P., Kestemont P., Milla S., Marie M. Effects of the thermal threshold and the timing of temperature reduction on the initiation and course of oocyte development in cultured female of Eurasian perch Perca fluviatilis. Aquaculture (2013), 376:90-96.
2. Trabelsi A., Gardeur J-N., Teletchea F., Brun-Bellut J., Fontaine P. Hatching time effect on the intra-spawning larval morphology and growth in Northern pike (Esox lucius L.). Aquaculture Research (2013), 44(4):657-666.
3. Teletchea, F., Fontaine, P. Levels of domestication in fish : implications for the sustainable future of aquaculture. Fish and Fisheries, first online publication 2012.
4. Castets M-D., Schaerlinger B., Silvestre F., Gardeur J.-N., Dieu M., Corbier C., Kestemont P., Fontaine P. Combined analysis of Perca fluviatilis reproductive performance and oocyte proteomic profile. Theriogenology (2012), 78(2):432-442.
5. Abdulfatah A., Fontaine P., Kestemont P., Gardeur, J-N., Marie M. Effects of Photothermal kinetic and amplitude of photoperiod decrease on the induction of the reproduction cycle in female Eurasian perch Perca fluviatilis. Aquaculture (2011), 322:169-176.
6. Teletchea F., Fontaine P. Particularities of early life stages in temperate freshwater fish species: comparison with marine species and implication for aquaculture practices. Aquaculture Research (2011) 42(5):630-654.
7. Trabelsi A., Gardeur J-N., Teletchea F., Fontaine P. Effects of 12 factors on burbot Lota lota (L., 1758) weaning performances using fractional factorial design experiment. Aquaculture (2011) 316:104-110.
8. Teletchea F. Fontaine P. Comparison of early-life stage strategies in 65 European freshwater fish species: trade offs are directed towards first –feeding of larvae in spring and early summer. Journal of Fish Biology (2010), 77(1):257-278.
9. Wang N., Teletchea F., Kestemont P., Milla S., Fontaine P. Photothermal control of the reproductive cycle in temperate fishes. Reviews in Aquaculture (2010), 2(4):209-222.
10. Taranger G.L., Carillo M., Schultz R.W., Fontaine P., Zanuy S., Felip A., Weltzien F.A., Dufour S., Karlsen O., Norberg B., Andersson E., Hansen T. Control of puberty in farmed fish. General Comparative Endocrinology (2009), 165:483-515.
11. Teletchea F., Fostier A., Kamler E., Gardeur J-N., Le Bail P-Y., Jalabert B., Fontaine P. Comparative analysis of reproductive traits in 65 freshwater fish species: application to the domestication of new fish species. Reviews in Fish Biology and Fisheries (2009), 19:403–430.
12. Teletchea F., Gardeur J-N., Kamler E., Fontaine P. The relationship of oocyte diamater and incubation temperature to incubation time in temperate freshwater fish species. Journal of Fish Biology (2009), 74:652-668.
13. Teletchea F., Gardeur J-N., Psenicka M., Kaspar V., Le Doré Y., Linhart O., Fontaine P. Effects of four factors on the quality of male reproductive cycle in pikeperch Sander Lucioperca. Aquaculture (2009), 291(3-4):217-223.
14. Teletchea F., Fostier A., Kamler E., Gardeur J.N., Le Bail P-Y., Jalabert B., Fontaine P. Comparative analysis of reproductive strategies of European fishes: application to the domestication of species in aquaculture. Cybium (2008), 32:300-302.
15. Teletchea F., Fostier A., Le Bail P-Y., Jalabert B., Gardeur J-N., Fontaine P. Storefish: A new database dedicated to the reproduction of temperate freshwater teleost fishes. Cybium (2007), 31 (2):227-235.
Neuroendocrinology and physiology of fish life cycles
Research Unit «Biology of Aquatic Organisms and Ecosystems » (BOREA) Sorbonne Universités – CNRS (MNHN, Paris, France) and Conservatoire National du Saumon Sauvage (Chanteuges, France)
Contact
Dr. Sylvie Dufour
sylvie.dufour@mnhn.fr
Phone : +33 1 40 79 36 12
Dr. Karine Rousseau
karine.rousseau@mnhn.fr
Phone : +33 1 40 79 36 14
Dr. Nédia Kamech
nedia.kamech@upmc.fr
Phone : +33 1 40 79 36 11
MNHN, UMR BOREA,7 rue Cuvier, CP32
75231 Paris Cedex 05, France
Patrick Martin
p.martin@cnss.fr
Phone : +33 4 71 74 05 45
Conservatoire national du saumon sauvage
Larma
43300 Chanteuges
Group description
The Research Unit BOREA « Biology of Aquatic Organisms and Ecosystems » (Director : Sylvie Dufour) is composed of 7 teams and aims at investigating evolutionary biology and ecology of aquatic organisms. The objective is to understand the origin, the role and the evolutionary mechanisms of aquatic biodiversity (from molecules to ecosystems) and to contribute predicting the responses to global, anthropogenic and climatic, changes. A large array of biological models are studied. Models are chosen for their phylogenetic position, biological cycle, ecological and economic relevance. The team S. Dufour/N. Kamech/K. Rousseau investigates the evolution of neuroendocrine systems and their roles in the control of the major steps of biological cycles (puberty, reproduction, metamorphoses, migration) with a special focus on teleost species.
The CNSS « Conservatoire National du Saumon Sauvage » (Director : Patrick Martin) is the largest restocking salmon aquaculture farm in Europe. It aims at ensuring the conservation of the unique Loire-Allier salmon population and at contributing to the restocking of other European long rivers (Rhin, Meuse). CNSS also participates in various research programs on the genetical and physiological characterization of Loire-Allier smolts, on the determination of restocking efficiency and on the modalities of migration in the Loire.
Main lines of research
-Evolution of neuroendocrine systems in metazoa;
-Physiological and neuroendocrine control of major steps of biological cycles (puberty, reproduction, metamorphoses and migration) in teleosts;
-Conservation of endangered species/populations.
Links
http://borea.mnhn.fr/
www.saumon-sauvage.org/
Relevant publications
1. Maugars, G., and Dufour. 2015. Demonstration of the Coexistence of Duplicated LH Receptors in Teleosts, and Their Origin in Ancestral Actinopterygians. PLoS One 10: e0135184. doi:10.1371/journal.pone.0135184.
2. Morini, M., Pasquier, R. Dirks, G. van den Thillart, J. Tomkiewicz, K. Rousseau, S. Dufour, and A.-G. Lafont. 2015. Duplicated leptin receptors in two species of eel bring new insights into the evolution of the leptin system in vertebrates. PLoS One 10: e0126008. doi:10.1371/journal.pone.0126008.
3. Rousseau, K., Dufour, and H. Vaudry. 2015. Editorial: A Comparative Survey of the RF-Amide Peptide Superfamily. Front Endocrinol (Lausanne) 6: 120. doi:10.3389/fendo.2015.00120.
4. Pasquier, J., – G. Lafont, K. Rousseau, B. Quérat, P. Chemineau, and S. Dufour. 2014. Looking for the bird Kiss: evolutionary scenario in sauropsids. BMC Evol Biol 14: 30. doi:10.1186/1471-2148-14-30.
5. Pasquier, J., Kamech, A. – G. Lafont, H. Vaudry, K. Rousseau, and S. Dufour. 2014. Molecular evolution of GPCRs: Kisspeptin/kisspeptin receptors. J. Mol. Endocrinol. 52: 101-117.
6. Maugars, G., Dufour, J. Cohen-Tannoudji, and B. Quérat. 2014. Multiple thyrotropin β-subunit and thyrotropin receptor-related genes arose during vertebrate evolution.. PLoS One 9: e111361. doi:10.1371/journal.pone.0111361.
7. Jeng, S. – R., – S. Yueh, Y. – T. Pen, Y. – H. Lee, G. – R. Chen, S. Dufour, and C. – F. Chang. 2014. Neuroendocrine gene expression reveals a decrease in dopamine D2B receptor with no changes in GnRH system during prepubertal metamorphosis of silvering in wild Japanese eel. Gen Comp Endocrinol 206: 8-15. doi:10.1016/j.ygcen.2014.08.001.
8. Blanchet-Letrouvé I., Lafont A.-G., Poirier L., Baloche S., Zalouk-Vergnoux A., Dufour S., Mouneyrac C. 2013. Vg mRNA induction in an endangered fish species (Anguilla anguilla) from the Loire Estuary (France). Ecotoxicology and Environmental Safety, 97: 103-113.
9. Fontaine, R., Affaticati, K. Yamamoto, C. Jolly, C. Bureau, S. Baloche, F. Gonnet, P. Vernier, S. Dufour, Pasqualini C. 2013. Dopamine inhibits reproduction in female zebrafish (Danio rerio) via three pituitary D2 receptor subtypes. Endocrinology 154: 807-18. doi:10.1210/en.2012-1759.
10. Imbert, H., Martin, P., Rancon, J., Graffin, V., and Dufour S. 2013. Evidence of late migrant smolt of Atlantic salmon (Salmo salar) in Loire-Allier system, France. Cybium 37, 1-2.
11. Pasquier, J., – G. Lafont, H. Tostivint, H. Vaudry, K. Rousseau, and S. Dufour. 2012. Comparative evolutionary histories of kisspeptins and kisspeptin receptors in vertebrates reveal both parallel and divergent features. Front Endocrinol (Lausanne) 3: 173. doi:10.3389/fendo.2012.00173.
12. Henkel C.V., Burgerhout E., de Wijze D.L., Dirks R.P., Minegishi Y., Jansen H.J., Spaink H.P., Dufour S., Weltzien F.A., Tsukamoto K., van den Thillart G.E.E.J.M. 2012. Primitive duplicate hox clusters in the European Eel’s genome. PLoS ONE 7(2): e32231. doi:10.1371/journal.pone.0032231.
13. Pasquier, J., – G. Lafont, S. – R. Jeng, M. Morini, R. Dirks, G. van den Thillart, J. Tomkiewicz, H. Tostivint CF Chang., K. Rousseau K., Dufour S. 2012. Multiple kisspeptin receptors in early osteichthyans provide new insights into the evolution of this receptor family. PLoS One 7: e48931. doi:10.1371/journal.pone.0048931.
14. Aroua, S., Maugars, S. – R. Jeng, C. – F. Chang, F. – A. Weltzien, K. Rousseau, and S. Dufour. 2012. Pituitary gonadotropins FSH and LH are oppositely regulated by the activin/follistatin system in a basal teleost, the eel. Gen Comp Endocrinol 175: 82-91. doi:10.1016/j.ygcen.2011.10.002.
15. Martin, P., Rancon, J., Segura, G., Laffont, J., Boeuf, G. and Dufour, S. 2012. Experimental study of influence of photoperiod ans temperature on the swiming behavior of hatchery-reared Atlantic salmon (Salmo salar) Aquaculture 362, 200-208.